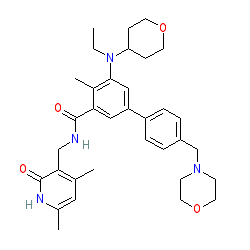
ANTICANCER AGENT “TAZVERIK® TABLETS 200mg” (TAZEMETOSTAT HYDROBROMIDE) LAUNCHED IN JAPAN FOR EZH2 GENE MUTATION-POSITIVE FOLLICULAR LYMPHOMA | News Release:2021 | Eisai Co., Ltd.

Cancers | Free Full-Text | Evaluation of Tazemetostat as a Therapeutically Relevant Substance in Biliary Tract Cancer
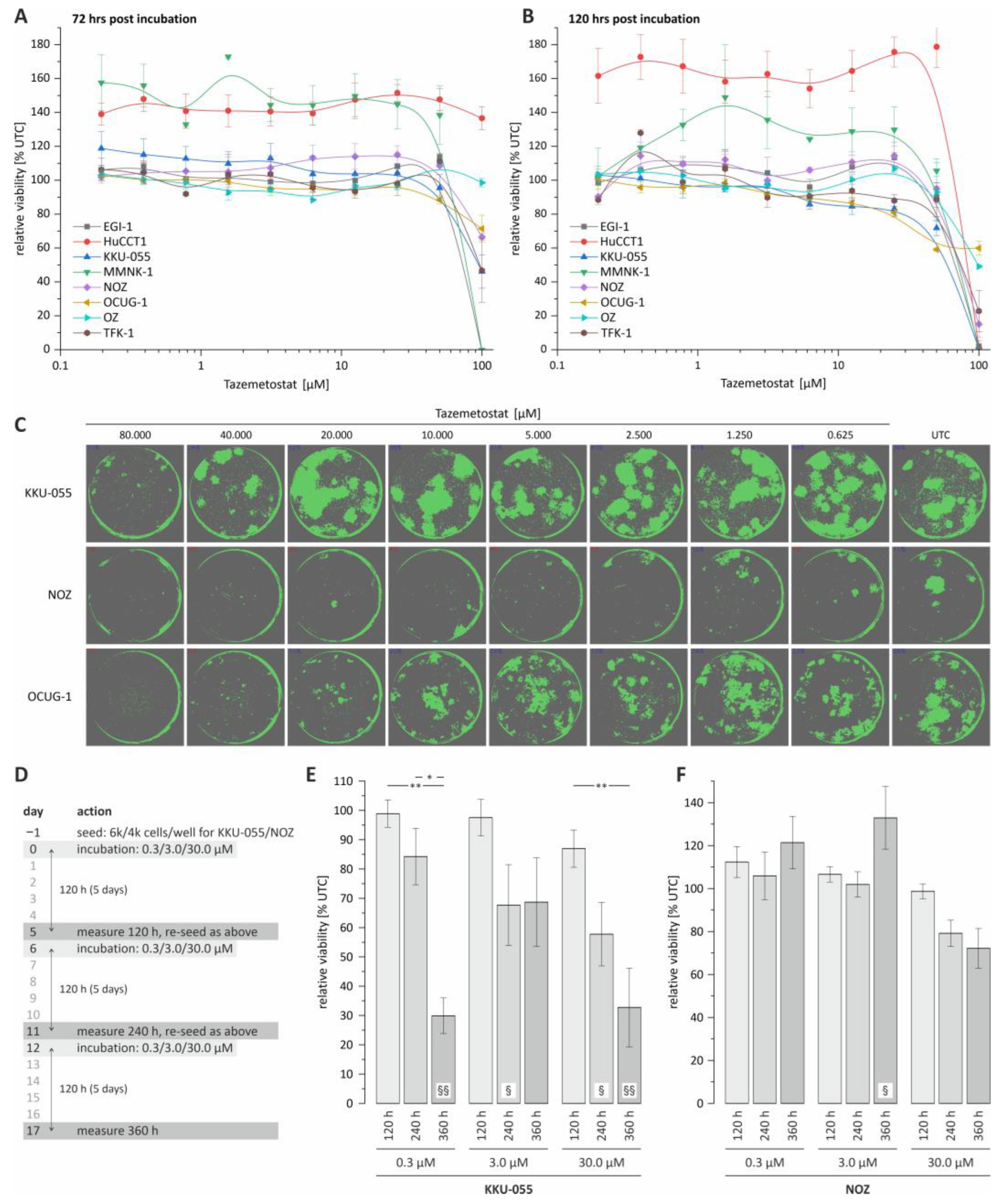
Cancers | Free Full-Text | Evaluation of Tazemetostat as a Therapeutically Relevant Substance in Biliary Tract Cancer

Targeting Enhancer of Zeste Homolog 2 for the Treatment of Hematological Malignancies and Solid Tumors: Candidate Structure–Activity Relationships Insights and Evolution Prospects | Journal of Medicinal Chemistry

Discovery of Dual Lysine Methyltransferase G9a and EZH2 Inhibitors with In Vivo Efficacy against Malignant Rhabdoid Tumor | Journal of Medicinal Chemistry

Testing the Addition of Tazemetostat to the Immunotherapy Drug, Pembrolizumab (MK-3475), in Advanced Urothelial Carcinoma
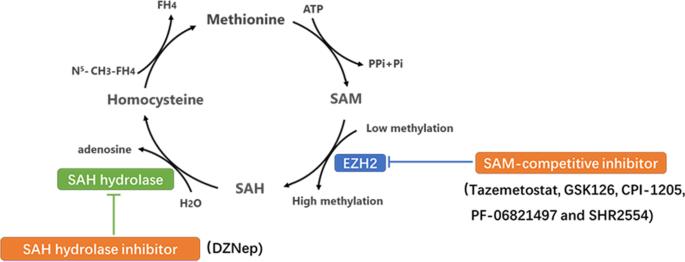
Finding an easy way to harmonize: a review of advances in clinical research and combination strategies of EZH2 inhibitors | Clinical Epigenetics | Full Text

Discovery of a Novel Covalent EZH2 Inhibitor Based on Tazemetostat Scaffold for the Treatment of Ovarian Cancer | Journal of Medicinal Chemistry

Epizyme Announces the U.S. Food and Drug Administration Lifts Partial Clinical Hold on Tazemetostat Clinical Program | Business Wire